Arcutis ZORYVE Infant Data Adds New Dimension To Atopic Dermatitis Thesis
- Arcutis Biotherapeutics reported positive Phase 2 topline results for ZORYVE cream in infants with atopic dermatitis.
- The study focused on a vulnerable pediatric group that currently has limited treatment options.
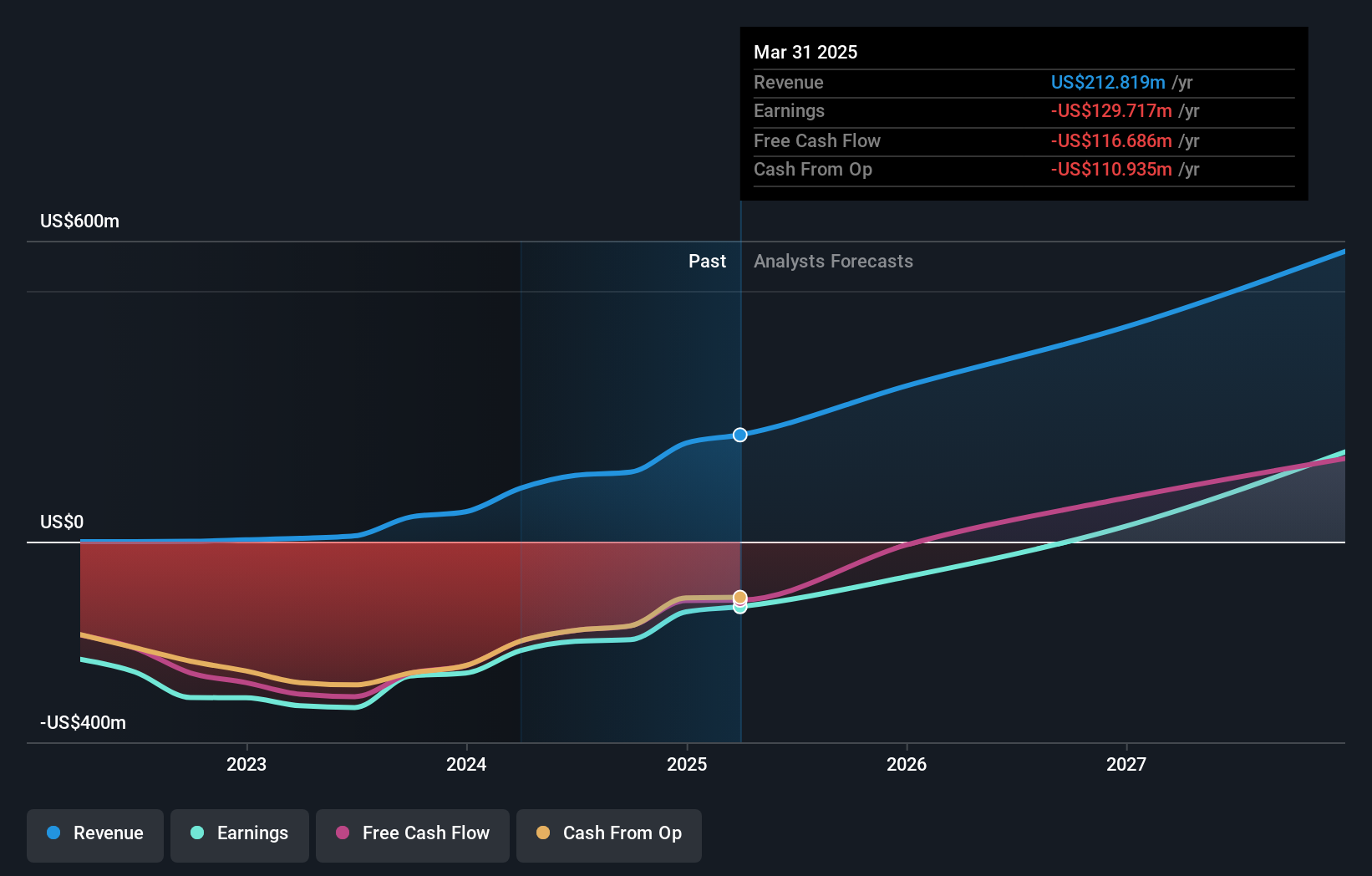
For investors watching Arcutis Biotherapeutics, NasdaqGS:ARQT, the latest ZORYVE update adds new context to a share price of $26.07. The stock has returned 109.9% over the past year and 57.7% over the past 3 years, while the 5 year return stands at a 25.1% decline. Short term moves have been more mixed, with a 1.3% gain over the past week and a 10.8% decline over the past month.
The new data in infants fits into an existing dermatology pipeline story that the market has already been pricing, positively at the 1 year level and more unevenly over longer periods. Investors will likely watch how Arcutis progresses ZORYVE toward potential regulatory discussions and how this pediatric indication fits alongside other priorities for NasdaqGS:ARQT.
Stay updated on the most important news stories for Arcutis Biotherapeutics by adding it to your watchlist or portfolio. Alternatively, explore our Community to discover new perspectives on Arcutis Biotherapeutics.
How Arcutis Biotherapeutics stacks up against its biggest competitors
The positive Phase 2 INTEGUMENT INFANT readout expands ZORYVE’s footprint into one of the most treatment-constrained segments of atopic dermatitis, infants aged 3 to under 24 months. A 58% EASI-75 response rate at Week 4 and a safety profile described as mild to moderate in terms of adverse events suggest ZORYVE could offer dermatologists a non-steroidal, once-daily option where long term steroid use is often a concern, which matters in a market that also includes players like Sanofi and Regeneron with Dupixent and topical competitors such as LEO Pharma.
How This Fits The Arcutis Biotherapeutics Narrative
This update lines up closely with existing community and analyst narratives that focus on expanding ZORYVE across multiple age groups and indications in large, underserved dermatology markets. The Phase 2 infant data builds on prior pediatric work, supporting the view that Arcutis is trying to build a franchise around one topical PDE4 inhibitor across atopic dermatitis and other chronic skin conditions rather than a one off product story.
Risks And Rewards For Investors To Weigh
- Positive efficacy and tolerability in infants strengthens the case for broader ZORYVE adoption across pediatric dermatology if regulators ultimately clear this age group.
- Management’s plan to file a supplemental New Drug Application in Q2 2026, if executed as indicated, could add a new label segment that supports future prescription volume and payer engagement.
- Arcutis remains heavily reliant on the ZORYVE franchise, so any slower than expected uptake in this or other indications would have an outsized impact on revenue concentration risk flagged by analysts.
- Access and reimbursement in infants could depend on payer decisions and treatment guidelines, which may limit how quickly dermatologists shift away from existing options.
What To Watch Next
From here, the key checkpoints are Arcutis following through on the planned Q2 2026 filing, any updates from regulators on review timelines, and early commentary from dermatologists on how they might use ZORYVE in infants versus current topical steroids or systemic options. If you want to see how other investors are thinking about ZORYVE’s role in Arcutis’ long term story, have a look at the community narratives for Arcutis Biotherapeutics and compare this news to the wider thesis and risk views.
This article by Simply Wall St is general in nature. We provide commentary based on historical data and analyst forecasts only using an unbiased methodology and our articles are not intended to be financial advice. It does not constitute a recommendation to buy or sell any stock, and does not take account of your objectives, or your financial situation. We aim to bring you long-term focused analysis driven by fundamental data. Note that our analysis may not factor in the latest price-sensitive company announcements or qualitative material. Simply Wall St has no position in any stocks mentioned.
Have feedback on this article? Concerned about the content? Get in touch with us directly. Alternatively, email editorial-team@simplywallst.com
 Nasdaq
Nasdaq Wall Street Journal
Wall Street Journal